Nickel(II) sulfate heptahydrate, for nickel plating, DIN 50970, for analysis
NICKEL SULFATE HEPTAHYDRATE
CAS: 10101-98-1
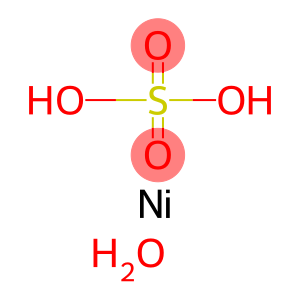
Molecular Formula: H14NiO11S
Nickel(II) sulfate heptahydrate, for nickel plating, DIN 50970, for analysis - Names and Identifiers
| Name | NICKEL SULFATE HEPTAHYDRATE |
| Synonyms | NickelSulphateAr NICKEL SULFATE HEPTAHYDRATE nickeloussulfateheptahydrate nickel(2+)sulfateheptahydrate NICKEL(II) SULFATE HEPTAHYDRATE NICKEL(II) SULFATE HEPTAHYDRATE, CRYST. Nickel(II)sulfateheptahydratecobaltfreegr Nickel(II) sulfate heptahydrate, for nickel plating, DIN 50970, for analysis |
| CAS | 10101-98-1 |
| EINECS | 600-153-9 |
| InChI | InChI=1/Ni.H2O4S.7H2O/c;1-5(2,3)4;;;;;;;/h;(H2,1,2,3,4);7*1H2/q+2;;;;;;;;/p-2 |
Nickel(II) sulfate heptahydrate, for nickel plating, DIN 50970, for analysis - Physico-chemical Properties
| Molecular Formula | H14NiO11S |
| Molar Mass | 280.86 |
| Density | 1.948g/mLat 25°C(lit.) |
| Boling Point | 330°C at 760 mmHg |
| Water Solubility | g/100g H2O: 26.2 (0°C), 37.7 (20°C), 50.4 (40°C) [LAN05]; soluble alcohol [HAW93] |
| Vapor Presure | 3.35E-05mmHg at 25°C |
| Appearance | Needle-Like Crystals or Powder |
| Color | Green to dark green |
| Merck | 13,6541 |
| Physical and Chemical Properties | Character Blue tetragonal crystal or green monoclinic crystal. The relative density is 2.07 The solubility is soluble in water, the aqueous solution is acidic, soluble in alcohol and ammonia. |
| Use | For electroplating, nickel batteries, catalysts and the preparation of other nickel salts, etc., and for mordant, metal colorants |
Nickel(II) sulfate heptahydrate, for nickel plating, DIN 50970, for analysis - Risk and Safety
| Risk Codes | R22 - Harmful if swallowed R40 - Limited evidence of a carcinogenic effect R42/43 - May cause sensitization by inhalation and skin contact. R50/53 - Very toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment. R68 - Possible risk of irreversible effects R48/23 - R38 - Irritating to the skin R20/22 - Harmful by inhalation and if swallowed. R61 - May cause harm to the unborn child R49 - May cause cancer by inhalation |
| Safety Description | S22 - Do not breathe dust. S36/37 - Wear suitable protective clothing and gloves. S60 - This material and its container must be disposed of as hazardous waste. S61 - Avoid release to the environment. Refer to special instructions / safety data sheets. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) S53 - Avoid exposure - obtain special instructions before use. |
| UN IDs | UN 3077 9/PG 3 |
| WGK Germany | 3 |
| RTECS | WT1157000 |
| HS Code | 28332400 |
Nickel(II) sulfate heptahydrate, for nickel plating, DIN 50970, for analysis - Upstream Downstream Industry
Nickel(II) sulfate heptahydrate, for nickel plating, DIN 50970, for analysis - Reference Information
| EPA chemical substance information | information provided by: ofmpeb.epa.gov (external link) |
| Introduction | nickel sulfate: one, two, four, six, seven hydrate and anhydrous, heptahydrate is a naturally occurring bisalum, green transparent crystal. Nickel sulfate heptahydrate loses six molecules of crystal water at 103 ℃, loses all crystal water at 280 ℃ and becomes anhydrous NiSO4, which is yellow-green crystal with specific gravity of 3.68, soluble in water, insoluble in ethanol and ether, it decomposes at 840 °c, loses sulfate, and becomes nickel oxide. |
| recovery preparation | a method for recovering copper and nickel from electroplating sludge, wherein step 1 separates copper is as follows: the leaching solution (hereinafter referred to as mother liquor) was adjusted to pH = 5-5.5 with sodium carbonate solution, and the precipitate was filtered to obtain mainly basic copper carbonate and copper hydroxide. The step 2 iron-nickel coprecipitation and iron are as follows: the mother liquor is adjusted to pH = 8-9 with sodium carbonate solution, and the precipitates are nickel carbonate, iron hydroxide and ferrous hydroxide, the precipitate was completely dissolved by addition of sulfuric acid and hydrogen peroxide, and then adjusted with alkali solution) pH = 5 the precipitate was filtered out, and the precipitate was iron hydroxide. The purification and separation of nickel products in step 3 are as follows: take the above filtered mother liquor and adjust the pH = 2 with sulfuric acid to enter hydrogen sulfide, convert the copper in the mixed solution into copper sulfide and separate it, the mother liquor in this case is a nickel sulfate mother liquor, and nickel sulfate heptahydrate can be produced by a method of concentrating crystallization. |
| Application | nickel sulfate heptahydrate can be used for electroplating, nickel battery, catalyst and preparation of other nickel salts, etc, metal colorants, etc. used in electroplating, nickel battery, catalyst and preparation of other nickel salts, etc., and used in printing and dyeing mordant, metal colorant, etc. used as hydrogenation catalyst, mordant, analytical reagent used in electroplating, nickel battery, catalyst and preparation of other nickel salts, etc., and used in mordant, metal colorant |
| preparation | dissolve the nickel sulfate to be refined in water, add excess concentrated ammonia, all of the nickel was converted into a nickel ammonia complex, and a small amount of hydrogen peroxide was further added thereto, followed by sufficient stirring, and the resulting Fe(OH)3 precipitate was removed by filtration. A concentrated NaClO4 solution was added to the filtrate, at which time Ni2 almost quantitatively produced a [Ni(NH3)6](ClO4)2 precipitate, which was filtered off and washed thoroughly with water. The precipitate was transferred to a beaker, pure sodium carbonate solution was added, and NH3 was evaporated by heating to form a nickel carbonate precipitate. After the mother liquor was separated by decantation, the precipitate was washed repeatedly with distilled water to obtain nickel carbonate as pure as possible. A pure nickel sulfate solution was obtained by dissolving nickel carbonate in dilute sulfuric acid. This solution is concentrated by evaporation and then crystallized at a temperature below 20 ° C. To give crystals of the heptahydrate NiSO4 · 7H2O. |
Last Update:2024-04-10 22:29:15
Supplier List
Multiple SpecificationsSpot supply
Product Name: Nickel(II) sulfate heptahydrate 99.999% trace metals basis Visit Supplier Webpage Request for quotationCAS: 10101-98-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: NICKEL SULFATE HEPTAHYDRATE Request for quotation
CAS: 10101-98-1
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 10101-98-1
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: Nickel(II) sulfate heptahydrate Request for quotation
CAS: 10101-98-1
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 10101-98-1
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Multiple SpecificationsSpot supply
Product Name: Nickel(II) sulfate heptahydrate 99.999% trace metals basis Visit Supplier Webpage Request for quotationCAS: 10101-98-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: NICKEL SULFATE HEPTAHYDRATE Request for quotation
CAS: 10101-98-1
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 10101-98-1
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: Nickel(II) sulfate heptahydrate Request for quotation
CAS: 10101-98-1
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 10101-98-1
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
View History
Raw Materials for Nickel(II) sulfate heptahydrate, for nickel plating, DIN 50970, for analysis
Downstream Products for Nickel(II) sulfate heptahydrate, for nickel plating, DIN 50970, for analysis


